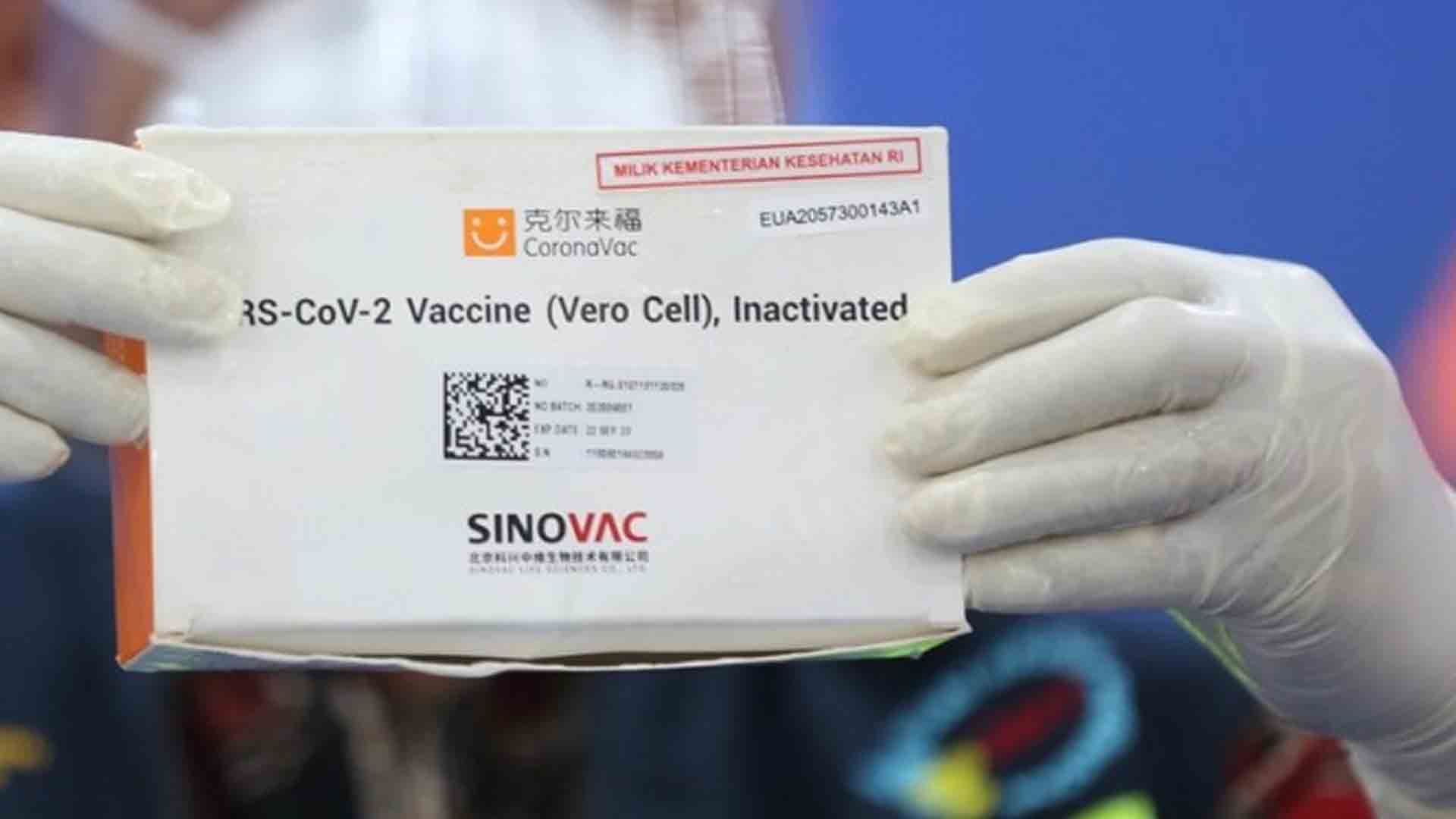
The Department of Health (DOH) and the Food and Drug Administration (FDA) on Wednesday allowed the use of CoronaVac-manufactured Sinovac for senior citizens, stressing vaccination may be done only after stringent evaluation of the person’s health status and exposure risk.
“After considering the recommendation of the experts and the current situation of high Covid-19 (coronavirus disease 2019) transmission and limited available vaccines, the FDA is allowing the use of Sinovac on senior citizens. Vaccination should be preceded by an evaluation of the person’s health status and exposure risk to assure that benefits of vaccination outweigh risks,” FDA Director General Domingo said in a statement.
The Department of Science and Technology’s Vaccine Expert Panel also recommended the use of the vaccine on the elderly.
While current efficacy data for senior citizens from Phase III trials is insufficient, the DOH and FDA noted “the benefits of using the vaccine for this particular group outweigh its risks and more scientific data on use for senior citizens may soon become available”.
Both agencies urged the senior citizens and persons with comorbidities to participate in the government’s Covid-19 vaccination program by registering in their respective local government units.
They also reiterated that registration for the vaccination is free. (PNA)